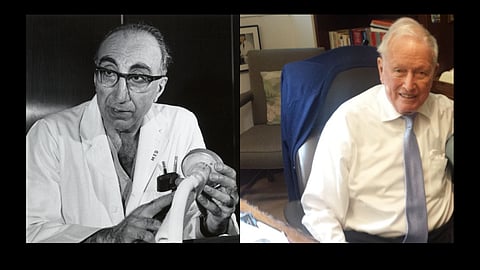
On April 4, 1969, inside an operating room at St. Luke’s Hospital in Houston, Dr. Denton Cooley implanted a device that had never before been used in a human. The patient, Haskell Karp, was in terminal heart failure. With no donor heart immediately available, Cooley and his team used a total artificial heart as a temporary measure.
The device had been developed in the laboratory of Dr. Michael DeBakey at Baylor College of Medicine with support from the National Heart Institute. Its use in humans had not been approved, and Dr. DeBakey was not informed of the procedure at the time.¹
The surgery was publicly announced soon after it was performed and received widespread media attention. A public appeal for a donor heart was made while the patient was supported on the device.⁴
The procedure became the first implantation of a total artificial heart in a human. It also triggered a conflict that would influence surgical ethics, research governance, and professional relationships for decades.
Dr. DeBakey and Dr. Cooley: The Surgeons Behind the Breakthrough