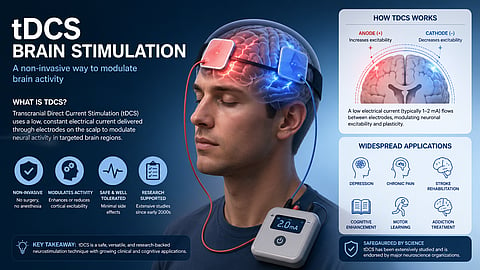
tDCS headsets are part of a rapidly developing space where neuroscience meets consumer technology. They do show promise, especially in areas like mental health and rehabilitation, but their ability to enhance everyday cognition is still not clear.
For now, it is better to approach them with awareness and a degree of caution, rather than assuming they will deliver dramatic results.
References
1. Nitsche, Michael A., Leonardo G. Cohen, Eric M. Wassermann, Alberto Priori, Nicolas Lang, Andrea Antal, Walter Paulus, Friedhelm Hummel, Paulo S. Boggio, Felipe Fregni, and Alvaro Pascual-Leone. 2008. “Transcranial Direct Current Stimulation: State of the Art 2008.” Brain Stimulation 1 (3): 206–23.
2. Neurolaunch. 2023. “TDCS Brain Stimulation: Exploring the Science and Potential of Direct Current Neuromodulation”.Accessed April 26, 2026.
3. Thair, H., A. L. Holloway, R. Newport, and A. D. Smith. 2017. “Transcranial Direct Current Stimulation (tDCS): A Beginner’s Guide for Design and Implementation.” Frontiers in Neuroscience 11 (November): 641.
4. Harvard Health Publishing. 2023. “Can Electrical Brain Stimulation Boost Attention, Memory, and More?” Harvard Medical School.
5. Kang, J., H. Lee, S. Yu, et al. 2024. “Effects and Safety of Transcranial Direct Current Stimulation on Multiple Health Outcomes: An Umbrella Review of Randomized Clinical Trials.” Molecular Psychiatry 29: 3789–3801.
6. Halo Neuroscience. 2024. “Neurostimulation Technology Overview.”